TBDC TsOH, Advanced Carbodiimide Coupling Agents for Peptide Synthesis in Aqueous and Green Solvents. Carbodiimide for minimal protection LPPS.
CAS: 3082189-36-1
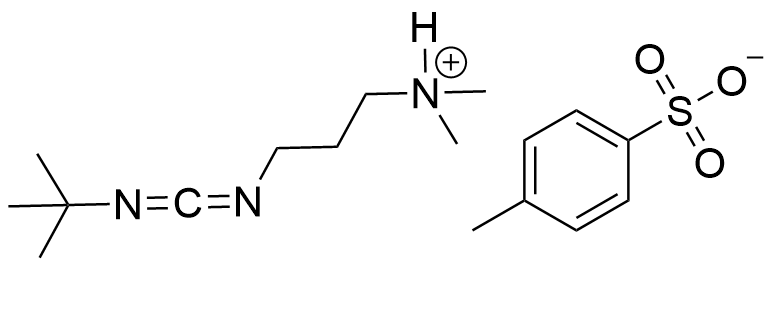
Synonym: 3-(((tert-butylimino)methylene)amino)-N,N-dimethylpropan-1-amine tosylate
Properties
Purity
≥98.0%
Molecular Formula
C17H29N3O3S
Molecular Weight
355,72 [gr/mol]
Appearance
White to off-white crystalline powder
Storage Conditions
Store in a cool place
Applications
TBDC (3-(((tert-butylimino)methylene)amino)-N,N-dimethylpropan-1-amine) salts represent a new generation of water- and organic-solvent-compatible coupling agents for peptide bond formation. These next-generation CDIs overcome key limitations of traditional reagents like EDC·HCl and DIC, offering:
- Minimal protection synthesis compatible. No need for protecting groups on His, Trp, Ser, Tyr, Thr
- High conversion (>95%) and low racemization (L:D > 95:5)
- Compatible with green solvents: EtOAc, 2-MeTHF, GVL, DMC
- Suitable for both manual and automated synthesis
- Forms open-chain stable intermediates unlike EDC
TBDC salts enable highly efficient amide bond formation in solution-phase, solid-phase, TAG – assisted LPPS ( TAPS) and green chemistry processes, eliminating the need for histidine and tryptophan side-chain protection in many cases.
Studies
Posters
TBDC® and QCD®: Novel water soluble carbodiimides for TAG assisted LPPS and minimal protection synthesis
Oleg Marder1, Srinivasa Rao Manne2, Hlobisile Nzama2, Beatriz G. de la Torre2, Fernando Albericio2, Alessandra Tolomelli3, Dario Corbisiero3, Lucia
Ferrazzano3, Nicola Patrian3 ,and Walter Cabri3